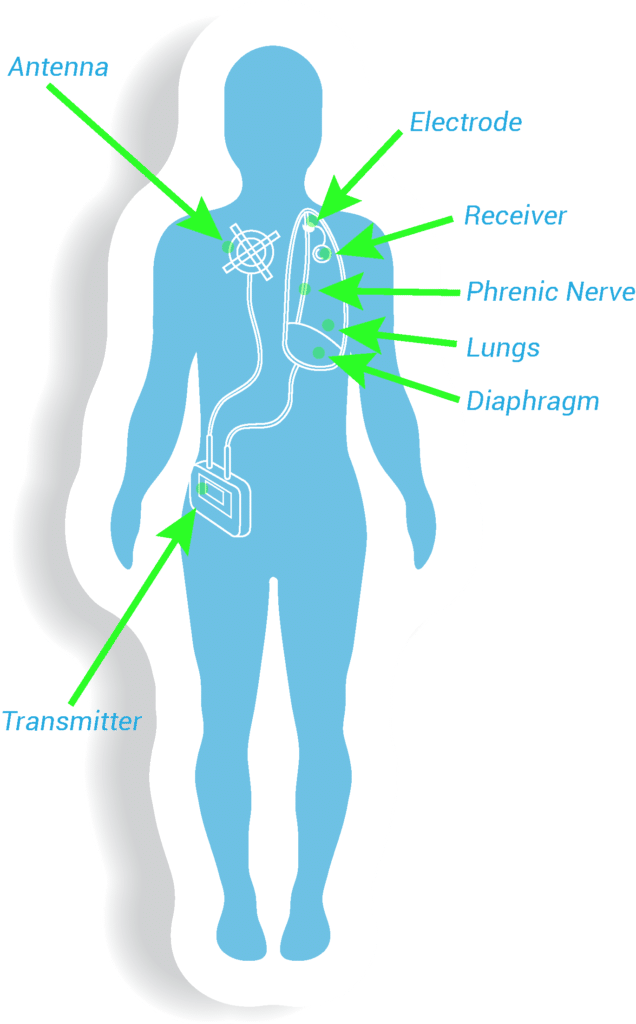
1Surgical Approach
Avery allows for either cervical or thoracic (thorascopic/VATS, robotic, uniportal VATS) approach whereas the competitor system requires several small incisions throughout the abdomen.
2 Adverse Events
3Reduced Infections
The Avery electrode is fully implantable and uses radiofrequency energy transmitted through intact skin to power the electrode. The competitor device has five wires protruding from the skin, necessitating ongoing wound care.
4Risk of Tissue Damage
There is risk of tissue damage with the competitor device because of the high operating voltage needed. The competitor manual states “Output voltages may approach 50 volt D. C. during operation.” The low voltage needed with the Avery device has been shown to not put tissue at risk for damage (J. of Pacing and Clinical Electrophysiology 23(6) June 2002)
5Risk of Intra-operative hypotension
Intra-operative hemodynamic instability can develop with the competitor device procedure because of the carbon dioxide introduced into the abdomen. (Anesthesia Intensive Care 2010; 38:740-743).
6Bilateral Redundancy
The Avery Diaphragm Pacemaker has two independent circuits, one for each side with its own controls, powered by its own battery and with its own antenna. If one side does not pace, the other continues pacing independently. The competitor device has one circuit and one battery for the entire system. If it stops operating, or if the battery is dead, neither side paces. The competitor manual states: “It is possible that stimulation from the diaphragm pacing system could stop either due to electrode breakage, cable disconnection, or stimulator failure. If one of these happens, breathing will stop. Without prompt attention, this could result in permanent disability or death.”
7Number of Anodes
The Avery Biomedical System has two implants, one for each side. Each implant has an electrode and its own anode, increasing redundancy. If one side fails, the other side continues to stimulate. The competitor device has four electrodes, but only one anode. If the anode lead wire is broken or does not make good contact, stimulation is not possible with any of the four electrodes.
8Alkaline Batteries
The Avery Diaphragm Pacemaker is powered by 4 inexpensive 1.5 Volt AA-size alkaline batteries available in any store anywhere in the world. These batteries last >400 hours when using factory default parameters. The competitor device is powered by a C-size 1.5 Volt alkaline battery. An expensive 3.6 Volt lithium battery can be used as an alternative. The manual states: “Replace the Alkaline battery every 96 hours of the NeuRx EPG use. This is about every 4 days if you are using NeuRx DPS full time”
9Parameter Adjustments
Avery Diaphragm Pacemaker has controls so that parameters can be adjusted by the patient (on/off, amplitude and respiratory rate). The competitor’s pacemaker has an on/off button without any possible adjustment of parameters. The patient cannot adjust the respiratory rate or the amplitude.
10Additional Equipment Needed
The Avery Diaphragm Pacemaker requires no additional equipment to adjust the parameters. The competitor device requires an expensive Clinical Station console necessary during the surgical procedure to stimulate the diaphragm to find the best location to implant the electrodes, used by the doctor to optimize the settings for comfort and effect, and used to program the desired settings into the device. (taken from company website)
11Maximum Stimulation Current
The Avery Diaphragm Pacemaker can generate a maximum stimulus current of 10 mA for each of the two implanted electrodes. Typical stimulus amplitude ranges from 1 mA to 5 mA. The competitor device can generate a maximum of 25 mA for each of the four implanted electrodes. Its minimum stimulus amplitude is 5 mA.
12Maximum Stimulation Voltage
The Avery Diaphragm Pacemaker generates a maximum voltage of 10 V. Typical stimulus voltage ranges from 1 V to 5 V. The competitor manual states: “output voltages may approach 50 volt D.C. during operation.”
13Cardiac Pacemaker
A cardiac pacemaker is not contraindicated in patients with an Avery Biomedical pacemaker. The competitor manual warning says “Do NOT use in patients with implanted electronic device (insufficient data is available, at this time, to establish safety with a cardiac pacemaker).” It also advises that “At this time, there is insufficient clinical data to determine safety in implanting patients with cardiac pacemakers, therefore, you should not be implanted with this device if you have a cardiac pacemaker or other implanted electrical device.”
14Long-term pacing safety
Long-term pacing with the Avery Diaphragm Pacemaker has been proven to be safe. There are patients pacing for more than 40 years. The manual of the competitor device advises that “The long-term effects of electrical stimulation of the diaphragm are unknown.”
15 Interference with the Heart
The Avery Diaphragm Pacemaker does not interfere with the heart rhythm. The manual of the competitor device states “there is a risk of cardiac arrhythmia being caused by the placement of the electrodes in the chest cavity.”