Press Release – June 10, 2005

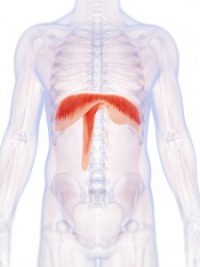
ABD remains committed to the excellent service and support of diaphragm pacing systems that our patients and doctors have come to expect. A diaphragm pacing system is an implanted phrenic nerve stimulator which can be used to free many quadriplegic and apneic patients from mechanical ventilation.
Breathing pacemakers are very cost efficient to operate and do not require the routine maintenance or variety of disposable supplies like a mechanical ventilator. Elimination of these costs can save over $20,000 every year for the life of the patient.
Our diaphragm pacing systems have full PMA approval from the US FDA and have qualified for the CE Mark under the European Active Implantable Medical Device Directive. Due to their high reliability and cost effectiveness, our devices are approved for Medicare reimbursement and by most private and government insurance plans.
The surgery is not complicated and is usually performed at a patient’s local hospital. Some implants can even be performed on an outpatient basis. Onsite engineering assistance is provided during surgery at no additional cost, and systems include transtelephonic monitoring (TTM) which allows the equipment to be monitored over a telephone from anywhere in the world.